
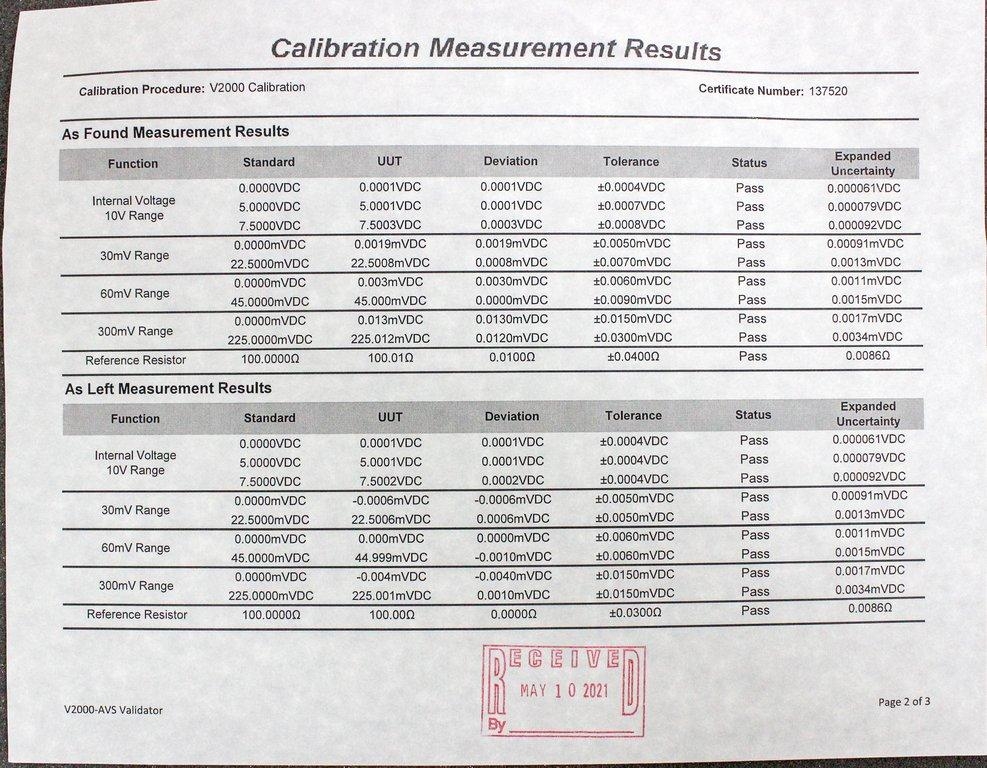









GE Kaye Validator X2010 Thermal Validation System Includes SIM Modules
GE / Kaye
Details
-
CategoryTesting Equipment
-
Dimension19.0in x 24.0in x 13.0in
-
Serial0012018
-
Shipping TypeFedEx Ground
-
ManufacturingDoes Not Apply
-
Voltage / Hertz120V / 60Hz US
Description
The GE Kaye Validator X2010 Thermal Validation System is a highly reliable solution designed for specific temperature and environmental monitoring in regulated industries such as pharmaceuticals, biotechnology, and medical device manufacturing. This unit, in good condition, appears to function well, powering on and passing basic tests. The Validator X2010 features modular input slots equipped to handle up to 36 separate inputs for thermocouple types T, J, K, offering a high resolution of 0.1°C. The validation system supports fast sampling of 8–12 channels/sec. It's especially suited for thermal mapping in autoclaves, ovens, and incubators, as well as for sterilization validation via lethality calculations. The system boasts secure software compliant with 21 CFR Part 11 for data integrity, including encrypted data, audit trails, and comprehensive reporting capabilities. The package includes three Sensor Input Modules (SIMs), storing calibration offsets and automating 2‑point/3‑point calibrations.
Technician comment
Includes: 3 sim modules
Key Features
- Modular Input Slots for up to 36 inputs
- High Resolution: 0.1°C with Type T
- Fast Sampling: 8-12 channels/sec
- CFR-11 Compliant Secure Software
- Supports T, J, K thermocouple types
- Includes 3 SIM modules
Specifications
- Input Channels: Up to 36 (3×12-ch)
- Thermocouple Types: T, J, K
- Resolution: 0.1°C, Type T 0.01°C
- Sampling Rate: 8-12 channels/sec
- Internal Memory: ~2 MB
- Battery Backup: ~30 minutes (NiCad)
- Connections: USB-Host, printer/PLC I/O
Weight
Imperial: 32.4 Pounds
Metric: 14.7 Kilograms
Shipping Dimensions
Imperial: 22.8 lb x 28.8 lb x 15.6 lb
Metric: 57.91 cm x 73.15 cm x 39.62 cm
Harmonized Code
9027.50
Harmonized Code Details
9027.50: Instruments and apparatus for physical or chemical analysis—for measuring or checking the temperature, very suitable for thermal validation systems in the pharmaceutical industry.
Product Enquiry
Have a question about this product? Send us your enquiry and our sales team will get back to you shortly.
Product Make Offer
Submit your best offer for this product. Our sales team will review your request and connect with you as soon as possible.
Comments 0
There are currently no public comments.